What Is a Certificate of Analysis for Research Peptides? COA Guide
A Certificate of Analysis, often called a COA, is one of the most important documents researchers can review when evaluating research peptides. It provides batch-specific documentation that may include product identity, purity percentage, testing method, molecular weight, batch number, and test date.
For research-use-only peptide materials, a COA helps researchers review documentation instead of relying only on product descriptions or general quality claims. A clear COA can support better traceability, product-to-document matching, and quality review.
This guide explains what a Certificate of Analysis means for research peptides, what details a peptide COA should include, how to review batch-specific documentation, and which red flags researchers should notice.
What Is a Certificate of Analysis for Research Peptides?
Quick Answer: Peptide COA Meaning in Simple Terms
COA Definition
A Certificate of Analysis for research peptides is a batch-specific document that summarizes testing and quality-related information. It may include product name, batch number, purity percentage, testing method, molecular weight, test date, and documentation details connected to a specific peptide material.
Why COA Documentation Matters in Research
COA documentation matters because it helps researchers connect a peptide material to specific testing information. Instead of reviewing only a product name or purity claim, researchers can look at the document that supports the batch being reviewed.
Key Documentation Terms
Important terms in a research peptide COA include Certificate of Analysis, COA, product name, batch number, lot number, purity percentage, testing method, molecular weight, test date, HPLC, LC-MS, and mass spectrometry.
What a COA Does and Does Not Show
A COA provides documentation for a specific material or batch. It can support quality review, traceability, and testing transparency. However, a COA is not a medical claim, cosmetic claim, therapeutic claim, or personal-use instruction.
Important Note
A COA is documentation. It should be reviewed as part of a research-use-only quality profile, not as a statement about human, veterinary, cosmetic, diagnostic, or therapeutic use.
What Information Should a Peptide COA Include?
Product Name
The product name on a COA should match the peptide material being reviewed. If the product page shows one name and the COA shows another, researchers may need clarification before relying on the document.
Check
The product name should match the product page, label, or product documentation.
Batch or Lot Number
The batch or lot number connects the COA to a specific production batch or documentation record. This is one of the most important details in research peptide documentation.
Check
The COA should be batch-specific. A generic COA without a batch or lot number is harder to evaluate.
Purity Percentage
Purity percentage describes the amount of target peptide detected in a tested sample under the stated analytical conditions. A purity value should be reviewed together with the testing method and batch details.
Check
Purity should be linked to a named testing method, not shown as an isolated number.
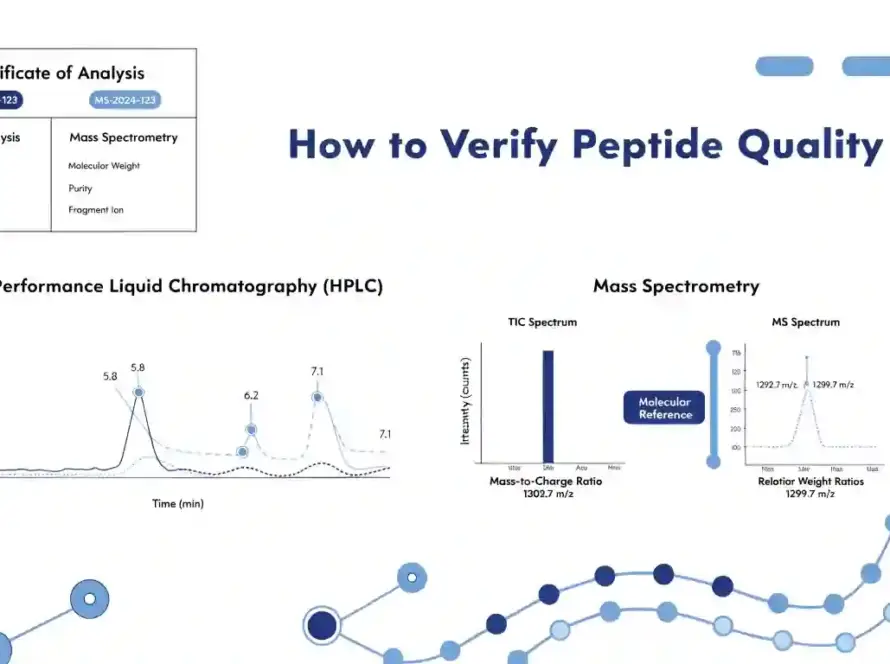
Testing Method
The testing method explains how the result was evaluated. Common methods that may appear in peptide documentation include HPLC for purity review and LC-MS or mass spectrometry for identity review.
Check
HPLC, LC-MS, or mass spectrometry should be listed where available and relevant to the result being reported.
Molecular Weight
Molecular weight information helps researchers review whether the documented material aligns with the expected peptide identity.
Check
Molecular weight should support identity review and match the expected peptide information where available.
Test Date
The test date shows when analysis was performed. A COA without a test date may be less useful because it does not show when the batch was reviewed.
Check
The COA should show when analysis was performed.
Why Batch-Specific COA Documentation Matters
Batch COA vs Generic COA
Batch-Specific COA Meaning
A batch-specific COA connects testing information to a particular product batch or lot. This helps researchers confirm that the documented purity, testing method, molecular weight, and test date apply to the material being reviewed.
How Batch Numbers Support Traceability
Batch numbers support traceability by linking a material to a specific documentation record. This makes it easier to compare the product page, COA, and testing information.
Quality Rule
The batch number should connect the product and COA. If the batch number is missing or does not match, the documentation may require further review.
Common Problems With Generic COAs
Generic COAs may provide broad information, but they are weaker than batch-specific documents. If the same COA is used across different products or lots, researchers may not know which material was actually tested.
Red Flag
The same COA appears across different products, different lots, or unrelated peptide materials.
How to Read a Research Peptide COA
Step 1: Confirm Product Identity
Start by checking whether the product name and molecular weight match the material being reviewed. Product identity should be consistent across the product page and COA.
Review
Review the product name, molecular weight, and documentation match.
Step 2: Match the Batch Number
Next, compare the batch or lot number on the COA with the batch information shown on the product page, label, or supporting documentation.
Review
Review the batch number, lot number, and product page details.
Step 3: Review Purity and Testing Method
A purity percentage should be reviewed together with the testing method. If a COA lists purity but does not show how it was evaluated, the result is less useful.
Review
Review purity percentage, HPLC, LC-MS, or mass spectrometry references where available.
Step 4: Check Test Date and Laboratory Details
Test dates and laboratory details can help researchers understand when analysis was performed and where the documentation originated.
Review
Review test date, laboratory reference, document readability, and authorization details where available.
COA Purity Percentage: What Researchers Should Understand
What Purity Percentage Means
Peptide Purity on a COA
Peptide purity on a COA refers to the percentage of the target peptide detected in a tested sample under specific analytical conditions. Researchers should review purity together with testing method, batch number, product identity, and documentation quality.
Why Purity Should Not Be Reviewed Alone
Purity is important, but it does not provide the full quality picture by itself. Researchers should also review molecular identity, batch information, test date, and product-to-document consistency.
Quality Rule
Purity needs method, batch, and identity context.
How Purity Fits Into Quality Verification
Purity is one part of the broader peptide quality verification process. It should be reviewed alongside COA details, HPLC references, identity information, and batch-specific records.
Related Guide
For a broader checklist, read our guide on how to verify peptide quality.
Testing Methods Commonly Found on Peptide COAs
HPLC for Purity Review
HPLC stands for High-Performance Liquid Chromatography. In peptide documentation, HPLC is commonly used to support purity review by separating and measuring components in a tested sample.
What HPLC Helps Show on a COA
HPLC can help support a reported purity percentage when the result is connected to the correct product, batch, and test date.
LC-MS or Mass Spectrometry for Identity Review
LC-MS and mass spectrometry are commonly used to support molecular identity review. These methods can help compare observed mass with expected molecular weight.
What Mass Spectrometry Helps Confirm
Mass spectrometry can help support whether the tested material aligns with the expected peptide identity.
Why the Testing Method Should Be Clearly Named
A named testing method gives researchers more context. Vague phrases like “tested” or “verified” are less useful without method details.
Red Flag
Be cautious when a COA or product page says “tested” without naming the method used.
COA vs Product Description: What Is the Difference?
What a Product Description Explains
A product description usually explains general product information such as name, quantity, category, or intended research-use-only positioning.
Product Page Role
Product pages give general product information and help researchers understand what material is being reviewed.
What a COA Confirms
A COA provides batch-specific testing documentation. It may include purity, testing method, molecular weight, batch number, and test date.
COA Role
COAs provide testing and documentation details connected to a specific batch or lot.
Why Researchers Should Review Both
COA vs Product Description
A product description explains general product information, while a COA provides batch-specific testing documentation. Researchers should review both because product pages help identify the material, and COAs help evaluate the supporting documentation.
Red Flags in Research Peptide COA Documentation
No Batch or Lot Number
A COA without a batch or lot number is harder to connect to the product being reviewed.
Red Flag
The COA cannot be matched to the product or batch.
No Testing Method Listed
A purity value without a named method gives researchers limited context.
Red Flag
Purity appears without analytical context.
No Test Date
A missing test date makes it harder to understand when the analysis was performed.
Red Flag
Analysis timing is missing.
Unreadable or Generic Document
A COA should be clear, readable, and specific enough to review.
Red Flag
The COA is not specific enough for documentation review.
Product Page and COA Do Not Match
If the product page and COA show conflicting information, researchers may need additional clarification.
Key Rule
Documentation should reduce uncertainty, not create more questions.
Research Peptide COA Review Checklist
Product Identity Checklist
Check Product Name, Molecular Weight, and Expected Identity
- Product name matches the product page
- Molecular weight is listed where available
- Identity details align with the expected peptide
- COA and product page refer to the same material
Batch Documentation Checklist
Check Batch Number, Lot Number, and Product Match
- Batch or lot number is visible
- Batch number matches the product documentation
- COA appears specific to the material being reviewed
- Generic or reused documents are avoided
Testing Information Checklist
Check Purity, Method, Test Date, and Lab Details
- Purity percentage is listed
- Testing method is named
- Test date is included
- Laboratory or authorization details appear where available
- Document is clear and readable
Final COA Review Checklist
Complete Peptide COA Checklist
A complete peptide COA checklist includes product name, batch or lot number, purity percentage, testing method, molecular weight, test date, laboratory details where available, document readability, and a clear match between the COA and product being reviewed.
How COAs Support a Documentation-Focused Peptide Catalog
Why Product-Level Documentation Helps Researchers
Product-level documentation helps researchers review each material more clearly. COAs, product pages, and batch details should work together as a documentation profile.
Catalog Review Point
Product pages and COAs should support each other with consistent information.
Why Research-Use-Only Labeling Still Matters
Research-use-only labeling keeps the product context clear. COA documentation should be reviewed within a research-use-only framework.
Compliance Note
Avoid human, veterinary, cosmetic, diagnostic, or therapeutic claims when discussing research peptide documentation.
Where COA Review Fits in Catalog Comparison
COA review is one step in comparing a documentation-focused peptide catalog. Researchers should also review product identity, purity information, batch records, testing references, and research-use-only positioning.
Research-Use-Only Materials
Researchers reviewing documentation-focused peptide materials can visit the
official Peptides Skin catalog.
Final Thoughts: Certificate of Analysis for Research Peptides
A COA Should Be Clear and Batch-Specific
A useful COA should clearly connect a peptide material to a specific batch or lot. This helps researchers review documentation with more confidence.
Key Point
Specific documentation is stronger than generic claims.
Purity Should Be Reviewed With Testing Context
Purity should be reviewed together with testing method, batch number, product identity, and test date.
Key Point
Purity needs method and batch information.
COA Review Supports Better Quality Verification
COA review is one part of peptide quality verification. It works best when combined with product-level documentation, testing references, and research-use-only labeling.
Key Point
COA review is one part of a broader peptide quality verification process.
Frequently Asked Questions About Research Peptide COAs
What is a Certificate of Analysis for research peptides?
A batch-specific testing document
A Certificate of Analysis for research peptides is a batch-specific document that summarizes testing information such as product name, purity, testing method, molecular weight, batch number, and test date.
What does COA stand for?
Certificate of Analysis
COA stands for Certificate of Analysis. It is a document used to provide testing and documentation information for a specific material or batch.
What should a peptide COA include?
Product name, batch number, purity, method, molecular weight, and test date
A peptide COA should include product name, batch or lot number, purity percentage, testing method, molecular weight, test date, and laboratory details where available.
Why is a batch number important on a COA?
It connects the document to a specific lot
A batch number is important because it connects the COA to a specific product lot or documentation record.
What does purity percentage mean on a COA?
It reports target peptide detected under testing conditions
Purity percentage reports the amount of target peptide detected in a tested sample under the stated analytical conditions.
What testing methods appear on peptide COAs?
HPLC, LC-MS, or mass spectrometry may appear
Peptide COAs may reference HPLC for purity review and LC-MS or mass spectrometry for molecular identity review.
Can a product description replace a COA?
No, product descriptions are not batch-specific testing documents
No. A product description provides general product information, while a COA provides batch-specific testing documentation.
What are red flags in a peptide COA?
Missing batch, method, date, or product match
Red flags include missing batch number, missing testing method, no test date, unreadable documentation, generic COAs, or product information that does not match the COA.
Research-Use-Only Disclaimer
Educational Purpose
This article is for informational research education only
This article is provided for educational and informational purposes in a research context.
No Human, Veterinary, Cosmetic, Diagnostic, or Therapeutic Use
Materials discussed should remain in a research-use-only context
The materials discussed are intended for research use only and are not for human use, veterinary use, cosmetic use, diagnostic use, or therapeutic use.